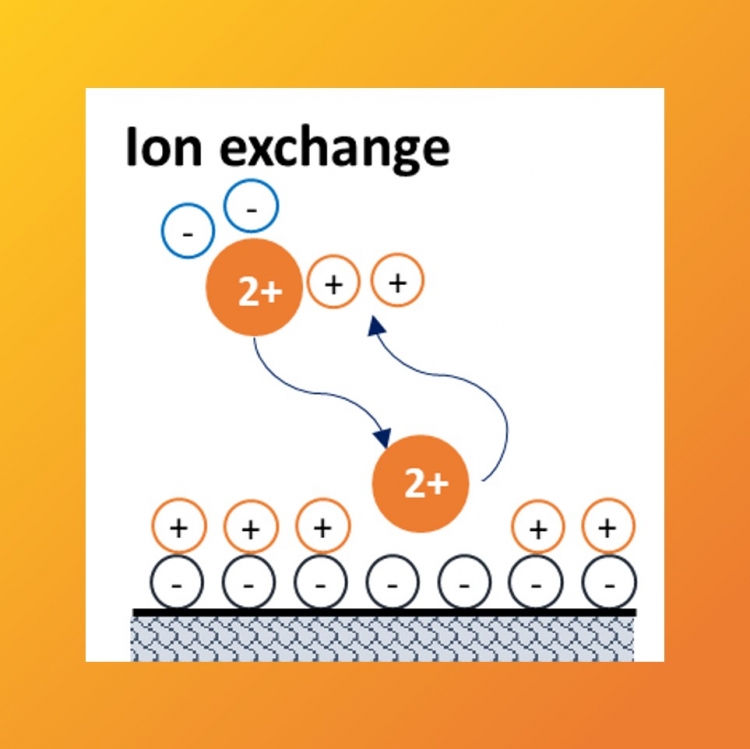
Ion-exchange chromatography is based on the differences in terms of electrostatic interactions of molecules regarding an adsorbent. Nowadays, industrial processes based on ion-exchange chromatography can be found in many different sectors from the food industry for sugars softening, to the biopharmaceutical industry for protein purification.
Modeling is widely used in certain fields of industry (aeronautics, automobile, oil & gas) to accelerate process development from R&D to industrial scale. Hence, the degree of adoption in biochemical process development is weak as, amongst other reasons, mathematical complexity of the approach is not well handled.
For simplification purposes, ion-exchange is quite often described with classical adsorption isotherms (Langmuir adsorption isotherms and alike) to simplify the problem and make the resolution of equations explicit. Although this can be a reasonable approximation in some cases, there are also numerous examples demonstrating the limits of this approach.
In industrial biotech (citric acid separation, amino-acids purification), complexity of ion exchange phenomena has been modeled and fairly predicted. However, as molecules holds a higher degree of complexity, they challenge the rigorous description of ion exchange phenomena involved in their isolation.
We will show how to deal with complexity of molecules and ion exchange to bring on hand modeling and consequently a rigorous and competitive design of bioprocesses. More precisely, we will show how an industrial process dealing with a mixture of complex multivalent enantiomers containing an active pharmaceutical ingredient (an antibiotic) can be better understood, optimized and adapted to changes (feedstocks) with a rational and methodological approach supported by modeling and simulation.
#chromatography #mechanistic #modeling #software #predictive #simulation #purification#antibiotic #ionexchange #adsorption