Downstream processing
Common problems
We help you overcome difficult separations to improve quality and yield while protecting sensitive molecules
Downstream processing often involves complex separations and purification steps that can significantly impact product quality, cost, and throughput. Typical challenges include:

Our solutions
Software license
Ypso-Ionic® was designed to speed up the development, optimization and scale-up of chromatographic processes. It combines 3 main functionalities, which can also be purchased independently:
SUPPORT PACKAGES
Our experts provide step by step guidance
from building a model with you to running simulations for you with the Essential,
Advanced and Comprehensive packages.
Collaborative partnership - Extended team
Our team of experts provides comprehensive downstream process development services. We work alongside your scientists and engineers, challenging assumptions and complementing internal initiatives. Acting as an extension of your team, we bring clarity by structuring data from diverse sources, testing scenarios with our models, and identifying the most relevant process levers.
Possible project goals include:
Our services let you benefit from the experience of our process experts and the capabilities of cutting-edge software tools built in-house. We propose three different packages:
EXPLORE, to get started with confidence: Set-up a first mechanistic model calibrated by Ypso-Facto's specialists and demonstrate its benefits on a concrete case study.
EXPAND, to widen tool capabilities: Take more physical phenomena into account, get more accurate predictions.
EXPLOIT, to support decision making: Capitalize on the obtained model to solve a real industrial challenge (e.g., reduce impurity content, improve yield, support filing, secure scale-up)
Custom software development and digital mirror
We have significant experience in developing scientific software tools for specific needs. These custom tools benefit from the solid foundations of our own software platform.
Furthermore, we offer a variety of web tools relying on the Ionic calculation engine to simulate specific processes with a user interface resembling the one of the machine, hiding all the details related to the model.
Such tools are particularly useful for training purposes and assess process robustness.
Digital Mirror
A digital mirror is a tailor-made virtual copy of your chromatographic process. It is designed for training, process representation, and simulation of "what-if" scenarios without running costly experiments.
What can be simulated:
What you receive:
Because the digital mirror is fully customized, you decide which information is critical and should be included.
Case studies
Process: Weak cation exchange chromatography
Product: Aminoglycoside antibiotic (Neomycin)
Challenge: A seemingly minor raw-material change in ion-exchange driven by supply, cost or sustainability pressures can unexpectedly disrupt separation performance and resin capacity, making deeper insight through smart experimentation and mechanistic modeling essential.
Achievements: Using a series of well-targeted experiments, a predictive mechanistic model was developed to describe the purification of Neomycin and the separation of its two isomers (B and C). The model accurately captured ion exchange mechanisms and acid–base equilibria, explaining two critical observations: the unexpectedly low resin capacity for isomer B and the strong influence of counterions on separation performance.
Comparing experimental and simulated breakthrough curves (Fig. 1), the model demonstrated excellent predictive capability across different feed compositions and operating conditions. It rationalized why the resin capacity for isomer B was four times lower than expected and predicted the failure of separation when switching from hydroxide to sulfate form (Fig. 2).
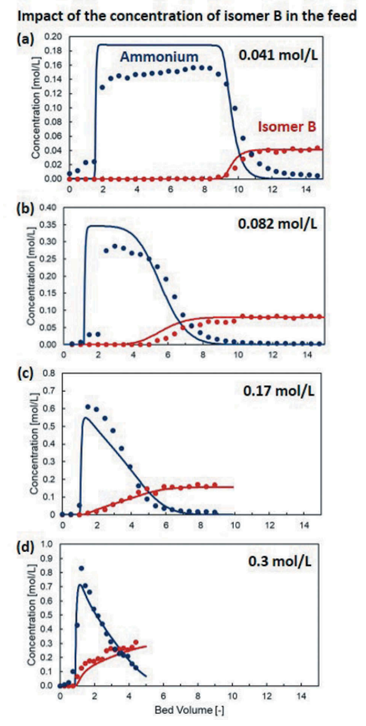
Fig. 1: Comparison between breakthrough curve experiments (symbols) and simulations (lines) on the impact of the concentration of Isomer B in the feed.
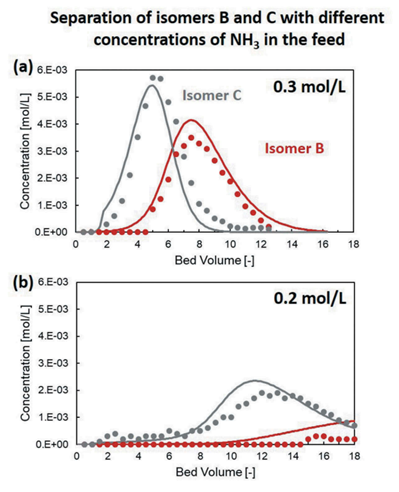
Fig. 2: Comparison between purification experiments (symbols) and simulations (lines) on the impact of the concentration of NH3 in the feed on the separation efficiency.
By leveraging this model, a wide range of scenarios could be assessed in silico, enabling optimization of operating conditions without extensive laboratory trials. This approach provided significant savings in time and material while ensuring robust process understanding.
Key message: Mechanistic modeling allows prediction of complex ion-exchange behaviors, reducing experimental burden and securing process performance for challenging separations.
Challenge: Industrial-scale separation of two sugars by multicolumn chromatography at high flow rates and feed concentrations suffers from poor efficiency, leading to excessive water consumption. Optimizing the operation conditions is critical to reduce resource consumption and overall costs.
Achievements: A web-based digital simulator was developed to make process optimization and operator training simple and accessible across the factory. Built on first principles, thermodynamics, kinetics, and fluid mechanics, the tool predicts process behavior and provides actionable insights without costly trials.
Key features:
- Real-time display of the process state in a dynamic flow sheet
- Real-time visualization of observable quantities like extract and raffinate purity and concentration and detailed flow compositions at specific points in the process
- Visualization of non-observable quantities like concentration profiles inside the chromatographic columns
- Automatic calculation of water use and KPIs such as yield and productivity
This easy-to-use platform empowers teams to explore “what-if” scenarios, identify optimal conditions, and improve efficiency. And the best is, it runs directly in your browser
Key message: A web-based simulator transforms multicolumn chromatography sugar separations into a transparent, controllable process, reducing resource consumption and boosting performance.
Challenge: A cosmetic active ingredient manufacturing process faced stricter specifications for the residual concentration of an odorous co-product. The objective was to meet these new requirements without compromising efficiency.
Methods:
- Identification of suitable technologies for removing the odorous co-product.
- Support for the customer in designing and executing an experimental program.
- Process simulations using mechanistic modeling to predict performance and optimize conditions.
Results:
- Successful design of an ion-exchange process and proof of concept with results exceeding expectations.
- Feasibility study completed for process scale-up.
- Technical and economic evaluation provided to guide implementation.
Key message: Mechanistic modeling combined with targeted experimentation enables efficient removal of challenging impurities and supports robust scale-up.
Antibiotics purification by ion-exchange
The power of mechanistic modeling and predictive simulation
Sometimes you have to change a raw material for various reasons (lack of availability, economic pressure, find alternative sustainable raw material), but this little change can impact significantly your process.
In the below article, you’ll see how some well-designed experiments and mechanistic modeling enable a tricky change of raw material in ion-exchange.
Changing the counter-anion on the raw material of this cation exchange process was expected to have very little influence on the separation. Yet, major issues were observed, including the loss of resin capacity.
Method: To address this challenge, a very complex system of 2 isomers with 6 pKas each was studied, rationalized, and modeled:
- With a set of smart experiments, the physico-chemical phenomena at stakes were understood and key parameters were measured
- The system was represented with a mechanistic ion-exchange model and simulated in our software Ypso-Ionic®.
Results: A new and efficient operating point was identified thanks to a rational scientific approach. A fully predictive IEX model was established.
This example illustrates the strength of using rigorous ion-exchange models, as opposed to the often-used classical adsorption models, to describe ion-exchange chromatography.
Further reading: Using mechanistic modeling for understanding antibiotics purification with ion-exchange chromatography
Your questions, answered
Not always. Many improvements can be achieved through process expertise and targeted experiments. Modeling becomes essential when complex separations, scale-up security, or precise control of critical quality attributes are required.
It captures the true physics and chemistry of the process, including ion exchange, hydrophobic interactions, and buffer effects. This allows prediction of separation performance, impurity clearance, and operating conditions without extensive laboratory trials.
Yes. Mechanistic models are based on physical principles, so they can be applied to different column sizes and configurations. This gives confidence when moving from lab to production scale and helps troubleshoot unexpected behaviors.
Ion exchange, hydrophobic interaction, reverse phase, affinity, and mixed-mode chromatography are standard. Custom models can be developed for specific needs.
Typically, a handful of well-designed experiments is enough to calibrate a first model. More experiments may be added later to refine accuracy.
Fewer experiments, faster development, improved yield and purity, and reduced material and buffer consumption.
Want to know more?
Don't hesitate to contact us!

