Upstream processing
Common problems
As global demand for Tide therapeutics grows, we collaborate with manufacturers to make solid-phase synthesis more efficient and reliable
Stepwise assembly of peptides and oligonucleotides on solid supports has enabled scalable and automated manufacturing. However, sequence-dependent variability and reaction kinetics as well as equipment characteristics introduce several process limitations.
These factors contribute to elevated material costs, longer production timelines, and inefficient use of environmentally sensitive reagents. Such constraints directly impact throughput and sustainability, especially on a manufacturing scale.

Our solutions
Collaborative Partnership - Extended Team
Our team of experts provides comprehensive upstream process development services. We work alongside your scientists and engineers, challenging assumptions and complementing internal initiatives.
Our experts can help to gain process understanding, accelerate your upstream development timelines, minimize empirical trial-and-error, assess the impact of multiple process parameters and enhance your process robustness from early-stage design through manufacturing.
We help you anticipate scale-up challenges such as resin swelling, solvent interactions, and equipment constraints, ultimately reducing risk, saving time and costs.
You can also rely on Ypso-Facto experts to build specific mechanistic models for your process. With our expertise and tools powered by the Ypso-Facto calculation engine, we can generate simulations of the synthesis cycles and identify the parameters to achieve your desired yields, visualize bottlenecks, or select the most efficient process for scale up.
Case studies
Real-world impact for peptide and oligo manufacturers.
Process: Solid-phase synthesis
Product: Oligonucleotide
Results: A comprehensive kinetic model of solid-phase oligonucleotide synthesis was developed, integrating mechanistic insights into the stepwise coupling, capping, oxidation, and detritylation reactions of SPOS. The model provides full dynamic results of the concentration evolution of all species in the liquid and in the solid phase and allows tracking the conversion of the oligonucleotide-resin towards the final product during each cycle (see Fig. 1, where On is the n-mer oligonucleotide resin). The formation of impurities along all the synthesis steps is predicted as well. To achieve this model, only one available laboratory experiment was used, no additional experiments were performed.
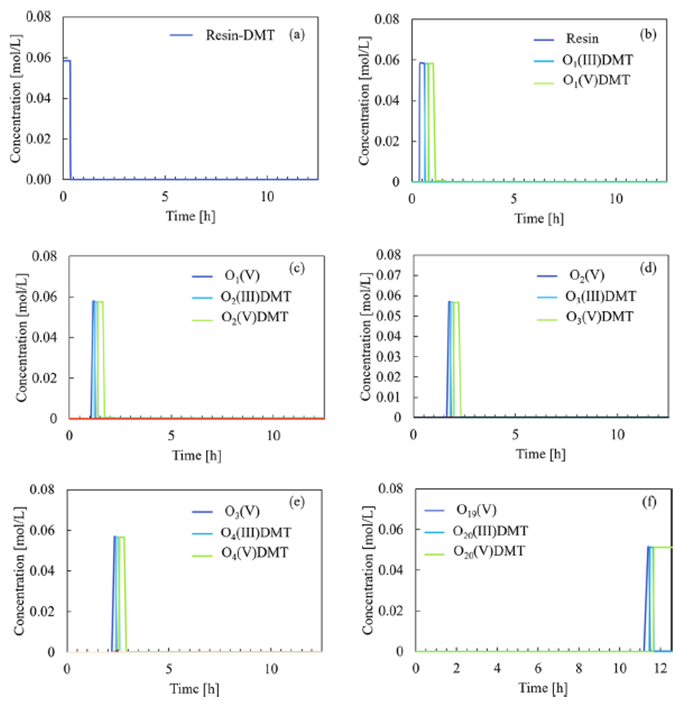
Fig. 1
Validation against experimental synthesis runs demonstrated that the proposed model can be used for predictive purposes with high accuracy, as readily illustrated by a comparison between the experimental and simulated N-X impurity profile (Fig. 2).
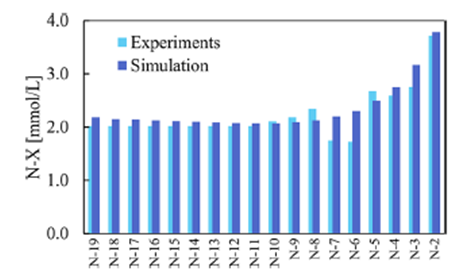
Fig. 2
Using a combination of predictive simulations and experimental data, rate-limiting steps and the influence of reaction conditions such as reagent concentrations, step durations, and stoichiometry on process performance were quantified (Fig. 3).
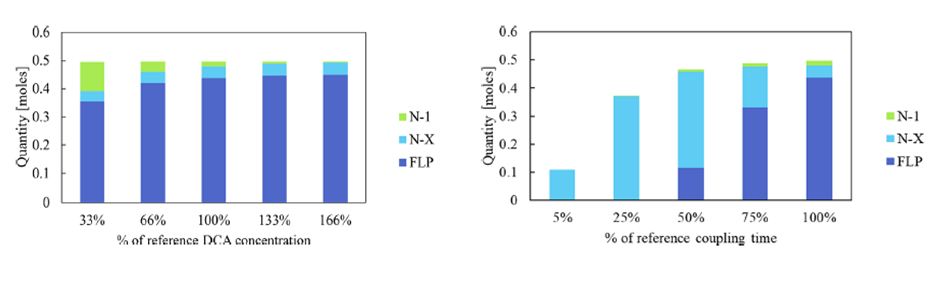
Fig. 3
The model can be used for process optimization, including adjusting cycle times and excess ratios. This quantitative framework is a practical application of a mechanistic model to improve solid-phase oligonucleotide synthesis with the objective of scalable production and cost-effective design of nucleic acid−based technologies.
Key message: solid phase synthesis can be optimized using a mechanistic model
Process: Solid-phase synthesis
Product: Peptide
Results: SPPS typically suffers from high solvent consumption associated with high cost and environmental burden. As reaching a specific maximum piperidine content after the end of deprotection is a critical process criterion, repetitive washing operations are performed to make sure to respect that specification under any circumstances. The potential for significant solvent consumption reductions while maintaining the desired piperidine concentration was realized by leveraging mechanistic modelling performed by Ypso-Facto.
Based on experimental data on the adsorption of piperidine on the peptide-resin, a predictive mechanistic model was calibrated, which allows simulation of different operating conditions for the washing step.
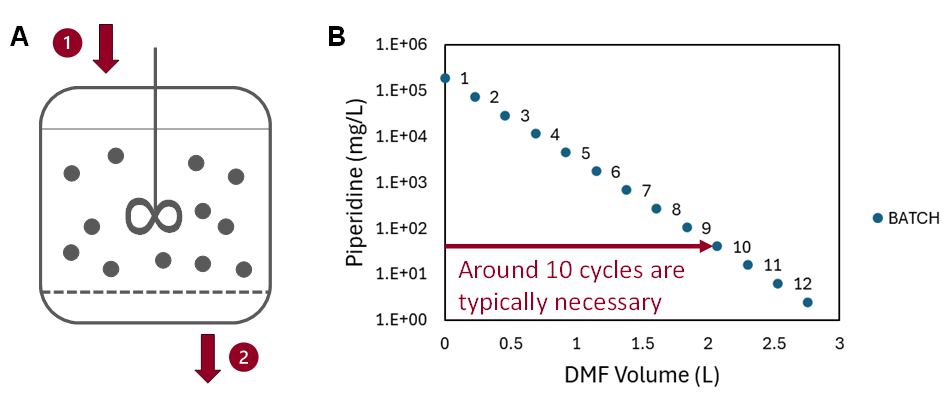
Fig. 1
Traditional batch washing (Fig. 1 A) consists of repetitive cycles of filling the reactor with solvent, stirring the resin-solvent suspension, and draining the solvent. The operation must typically be repeated 10 times for the piperidine content to drop below the desired 100 mg/L threshold.
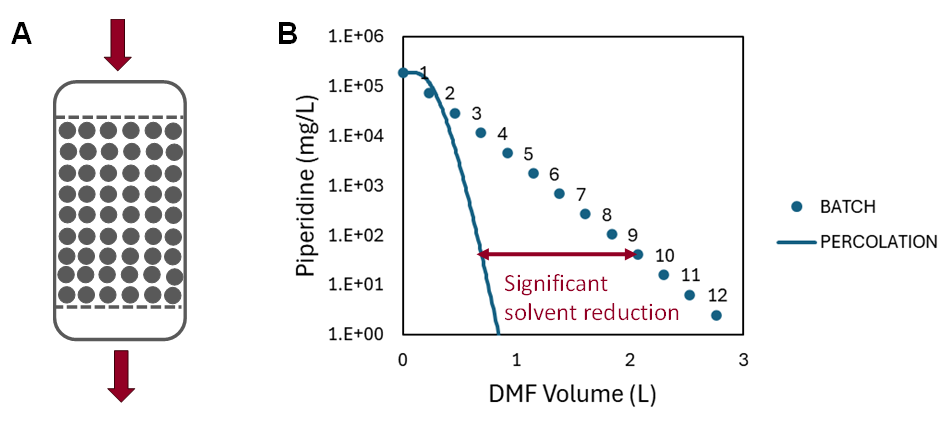
Fig. 2
Ypso-Facto suggested and modelled a different approach by using percolation washing. During percolation washing (Fig. 2 A), the resin bed is not stirred at all. The solvent is simply pushed through the column until the piperidine content at the outlet is sufficiently low. This method of operation allows a reduction of the solvent volume consumed for washing of 57% to reach the same piperidine content at the reactor outlet as during the batch washing.
Key message: mechanistic modeling can be used to investigate processes aiming at reducing solvent consumption.
Read more: Reduced solvent consumption by 57% in peptide solid phase synthesis
Your questions, answered
No, mechanistic modeling is not a prerequisite for optimizing and securing scale-up of peptide and oligonucleotide assembly processes. Whether or not a mechanistic model is required strongly depends on the objective that is to be reached. Many issues can be addressed thanks to the experience and engineering knowledge of our experts. The more highly precise and tightly controlled targets need to be met, the more building a mechanistic model makes sense.
With our models, we can simulate each stage of solid‑phase synthesis from deprotection, washing and coupling to capping and cleavage. This allows predicting yields, identifying bottlenecks, and optimizing conditions while reducing the need for extensive laboratory trials.
The number of required experiments strongly depends on the desired scope and precision of the predictive abilities of the model. A first model can be generated by using a handful of well-thought experiments and smart approximations. If great confidence in validity over a wide variety of operating conditions, determination of reaction endpoints and product purities is to be achieved, more experiments will be required to replace some approximations with high-quality training data for the model.
Want to know more?
Don't hesitate to contact us!
