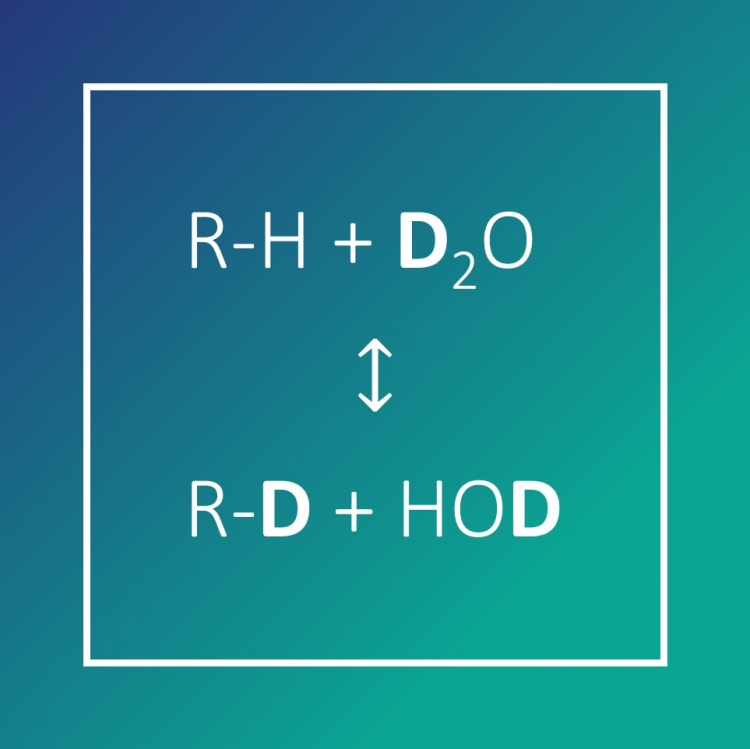
Ninety years after its discovery, Deuterium is in the spotlights. Since the beginning of the 21st century, a number of biotechnology companies have successfully explored its potential in drug candidates and generated intellectual property. This approach has been "validated" by the first approval by the FDA of a deuterated drug in 2017. This renewed interest has also triggered an acceleration of the development of new methods for the synthesis of deuterated drugs. In this webinar, we will first briefly go over the history of deuterium and its use over the years. We will then explore why it has been increasingly used in medicinal chemistry and how ‘’leaving the deuterium in the drug’’ can reduce the toxicity, the metabolization of a drug and overall improve its pharmacokinetic profile. We will then focus on the synthesis of deuterated compounds from the historical methods to the more recent ones and highlight the challenges that remain. Lastly, we will show how a chemical engineering approach could bring a better understanding and control of the deuteration reactions and allow for the large-scale production of complex deuterated drugs.
In this webinar, Thomas Jennequin presents insights on the use of Deuterium in medicinal chemistry and the advantages of having some deuterated positions on APIs. For instance, Deuterium can modify drug metabolization, pharmacokinetics, or toxicity.
He also highlights some of the challenges of the synthesis of d-molecules. Thomas also describes how process engineering can help understand and control the chemistry and support the production of these complex molecules at large scale.